 Let’s use the reaction between sodium chloride and silver nitrate as an example. The first step to writing a net ionic equation is balancing the chemical equation present. It makes it easy to see the active molecules in a reaction, since they are the only ones present in the equation! Writing net ionic equations In an ionic equation: Number of atoms of each elements must be balanced Total charges carried by the ions must be balanced (e.g. For example: CaCO3 is formed from CaCl2 and. You can use words for an ionic equation (either complete or net), it just isn't usually done. 
Ions (never words) will be used for the complete ionic equation and the net ionic equation, which will follow just below. Recall that ionic compounds that dissolved in water will dissociate completely into ions (have charge). The balanced ionic equation involving all the component ions of ionic compounds is called a balanced ionic equation. HCl(aq) + NaOH(aq) -> NaCl(aq) + H2O() Only full formulas (never words or ions) are involved in a complete molecular equation. Why do we use net ionic equations?Īs described above, we use net ionic equations to emphasize the molecules that undergo a change in the reaction. Complete ionic equations show all the atoms in a chemical equation whereas net ionic equations do not show the ions that cancel out from both sides of the eqn. Writing ionic equation is extremely similar to writing chemical equations. The net ionic equation is balanced for mass and charge and lists the state of matter of all species. For the net ionic equation, start with the complete ionic equation. In this equation, the spectator ions are not present. Ag + (aq) + Cl (aq) AgCl (s) The net ionic equation tell you at a glance which ions influence product formation and whether or not there is a solid present. The definition of a net ionic equation is an equation that depicts only the molecules or ions that are actively involved in the reaction or those that undergo a change. Answer (1 of 7): Writing Complete Ionic Equations When aqueous solutions of sodium phosphate and calcium chloride are mixed together, an insoluble white solid forms. Aqueous solution: a solution in which the solvent is water.(Only compounds that are aqueous are split into ions. 2Na (s) + 2HCl (aq) -> 2NaCl (aq) + H 2 (g) Step 2: Split the ions. Spectator ion: ions in a reaction that do not undergo any change. Solution: Step 1: Write the equation and balance it.Write down your observations molecular equation ionic equation and net. 
Only the aqueous ionic compounds (the two chloride salts) are written as ions: (8.5. They therefore appear unaltered in the full ionic equation. Balanced equation that has an equal number of atoms of each element on both sides of the equation is called a balanced chemical equation, i.e., the mass of the reactants is equal to the mass of the products. The chemical reaction: 8 Fe + S 8 FeS is a:Q1: What is an Ionic Equation. Solution The elemental metals (magnesium on the reactant side, copper on the product side) are neutral solids. Net ionic equation: a molecular equation that depicts the ions or molecules that experience a change within a reaction. The balanced equation for the reaction between HCHO and KOH is given by HCHO (aq) + KOH (aq) KCHO (aq) + HO (l) Ionic equation involves writing a balanced equation showing all the ions in a chemical reaction. The balanced equation for the reaction between iron III chloride and ammonium hydroxide is.Complete ionic equation: a molecular equation that separates the molecules into their ion forms. Net Ionic Equation shows only the ions participating in the chemical reaction, without the spectator ions. 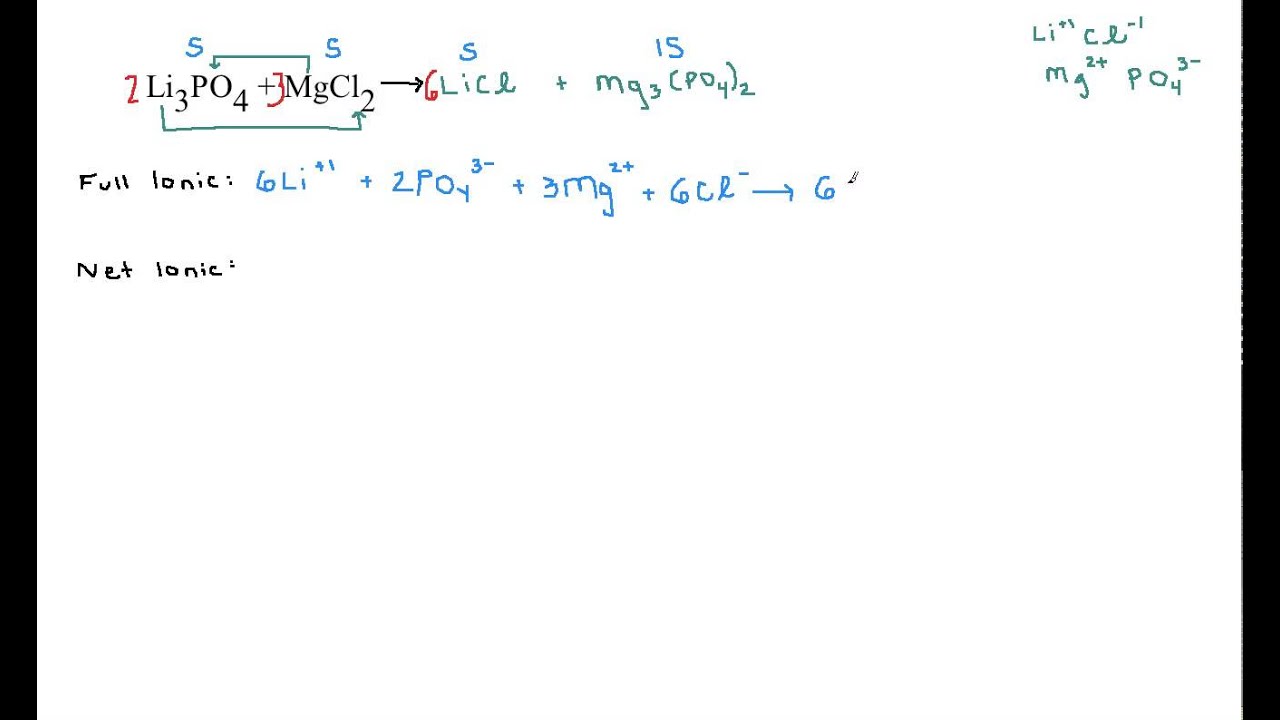
In addition, you will learn how they are useful for understanding which ions in the solution are active in a reaction and which are spectator ions. By gaining a hydrogen (and a unit of charge) the hydroxide ion transforms into a water molecule.In this tutorial, you will learn how to write net ionic equations. The mechanism of the reaction becomes more clear by inspecting the net ionic equation: the ammonia molecule is created from the ammonium ion when the hydroxide ion strips a hydrogen away from it. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
